Sustainability engineering - in short
A data-driven approach to reducing the environmental footprint of your future medical devices
Clear, actionable data on your product’s environmental impact supports your decision-making throughout the device development process: our iterative Life Cycle Screening method in combination with industry best practices and Ecodesign principles gives you a solid basis for making informed sustainability choices at every stage of development.

Sustainability engineering
Our capabilities
Our approach and team can help you with:
Standardization, modularization
and product architecture
Design Review
Design for Manufacturing
and Assembly (DFM & DFA)

Sustainability engineering - the long read
Gain clarity and assurance about the effect of your sustainability effort
The medical device manufacturers we work with are seeing a steep increase in sustainability demands coming at them from every angle. Despite a general wish to share in the environmental responsibility and future-proof their businesses, setting the sustainability effort on a steady trajectory can be a major challenge for MedTech manufacturers.
Development cycles lasting several years and strict regulatory requirements to products and safety can make it difficult to incorporate sustainability into an already complex development process.
Make informed decisions about sustainable design improvements
Our two-pronged approach provides a framework for making sustainability assessments and improvements from the very beginning and throughout the device development project. This is done by combining Ecodesign principles with Life Cycle Screenings and bringing important aspects from Life Cycle Assessments into the core of the MedTech development process in a manner that is much faster and more agile than with traditional LCAs.
Rather than being retrospective in character like LCAs, our approach offers value during all stages of product development. The goal is to bring a proactive approach to sustainable product development by gaining fast access to dependable sustainability data throughout the development process.
Ecodesign + Life Cycle Screenings = improved environmental impact
We see that using Ecodesign principles when deciding on sustainable design improvements helps our clients to concentrate their effort on the changes that have the most impact. The Ecodesign principles are firmly grounded in industry best practices end therefore align with the sustainability guidelines of our clients. The principles are also based on established theory from the Ecodesign research community, and they align with designing for a circular economy.
Our Life Cycle Screenings are based on the Life Cycle Assessment methodology in ISO 14040/14044 and provide similar information as an LCA, but based on generic data and assumptions. This makes it applicable during a product development process, providing invaluable feedback to the Ecodesign process in an iterative way. Whether your focus is sustainable packaging or medical device sustainability, our experts can help.
Working with sustainability data can seem a bit far removed from reality. Our clients value our development mindset and the fact that we are rooted in MedTech because it means our focus is on finding solutions that contribute real value to their development projects and ultimately support their business. The sustainability improvements we bring to the project have to be applicable in a device development context and at the same time be in alignment with our clients’ sustainability goals.”
– Martin Høier Thomsen, Senior Development Engineer & Sustainability Engineering

Want to know more?
Let's get in touch
As our lead on Sustainability Engineering, Martin is looking forward to meeting you and diving into how our approach can help you improve your sustainability. Send him a note and he will be happy to meet and present our insights.
White Paper
Sustainability Challenges
This white paper explores potential obstacles and barriers for making product development more sustainable as experienced by a broad sample of the Danish MedTech industry.
What we do
We have the expertise to take you from A to Z
Whether you need a single competency or a combination of skills, our experts are ready to assist you through the entire development process. Based on the specific needs of the project, we assemble the ideal team of specialists.
Since joining forces with the MGS Group, we are able to integrate advanced manufacturing knowledge into the earliest design phase and minimize the number of iterations. Our complete offering takes your project seamless through the entire value chain ensuring faster time-to-market combined with minimized risk. What’s not to like ?
related cases
Cases you might be interested in:

How an FDA pre-submission enhances your usability engineering process
Discover the advantages of incorporating FDA pre-submission insights into your usability engineering process for streamlined success.

Principles for Combination Products: Questions and Answers Guidance for Industry and FDA Staff
news from fda Application of Human Factors Engineering Principles for Combination Products — Questions and
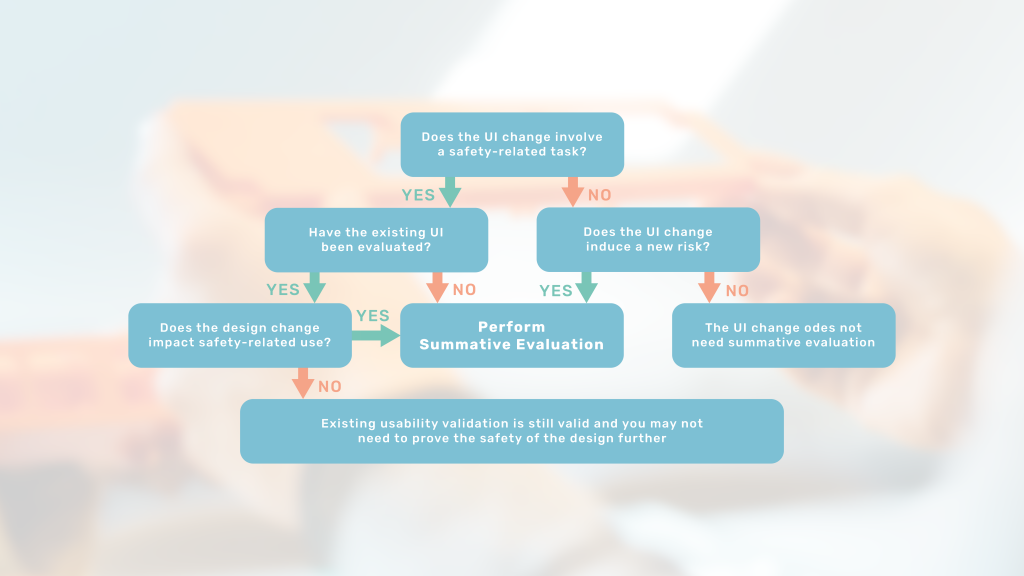
When is a new summative evaluation needed?
This article will introduce you to assessing the usability impact of your design change and thereby plan for any needed summative evaluation.
