ISO 14971:2019
Identifying potential use errors
When working with usability engineering and interface design, a very effective and systematic way to help you identify potential use errors is to use the ‘PCA analysis’. The PCA analysis breaks down each use step into three components: Perception, Cognition, and Action, hence the name PCA.
Breaking each step of the users interaction with the device down to these three components, will help you understand what each task requires from the user.
A use error or difficulty is likely to occur if the user is unable to meet one of these task requirements related to perception, cognition and action. In this way, the PCA analysis, can be a very usefull tool to help you identify use errors related to hazardous situations.
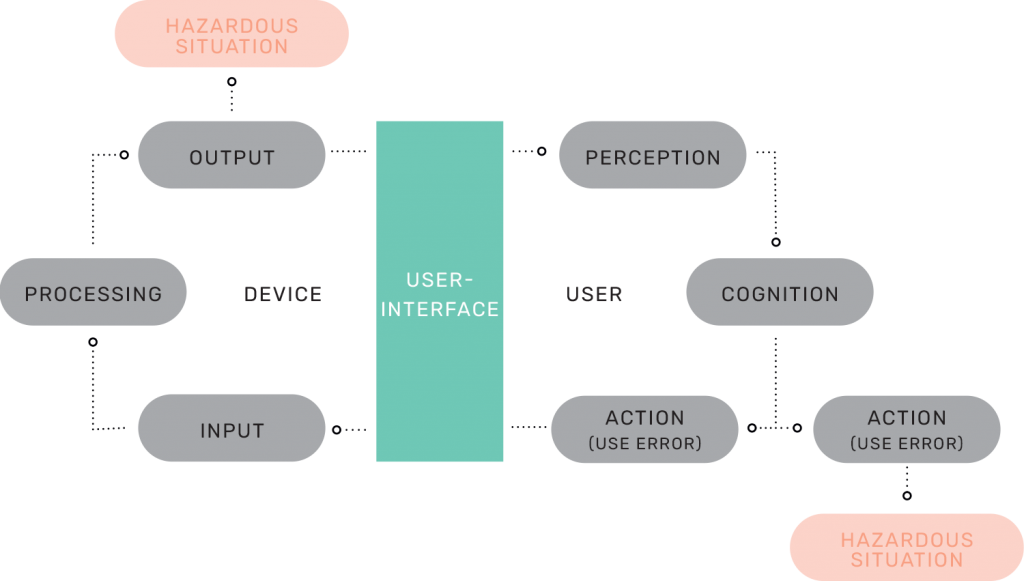
User-device interaction model
We have now looked at how steps in user tasks can be broken down to Perception, Cognition, and Action. The PCA analysis is derived from the above model of user-device interaction. The model illustrates a user interacting with a device through a user interface. The user gives input to the device (e.g. presses a button), which the device processes to provide an output back to the user through the device user interface.
The user shall be able to perceive this output from the device, understand what to do with the output and act accordingly. This model can be applied to consider sources of potential use errors or hazardous situations caused by the device output. Note that we differentiate between:
- hazardous situations caused by a response of the medical device and
- hazardous situations caused by a user action or lack of action based on information obtained from the medical device.
PCA analysis provides valuable input to the user interface design
We have introduced the PCA analysis and the user-device interaction model. Let’s consider a simple example of how to apply the PCA analysis in order to both (1) identify potential use errors and (2) identify design input requirements:
Task step
-
Power on device

P/C/A component
-
Perception – detect ON/OFF button
-
Cognition – understand that pressing the ON/OFF button powers on the device
-
Action – apply pressure to depress the ON/OFF button
Use error
-
The user cannot detect the button
-
The user does not understand that pressing the ON/OFF button powers on the device
-
The user is unable to apply enough pressure to depress the button
Consequence
-
Device is not powered on
Design input requirement
-
ON/OFF button shall be backlit
-
ON/OFF button shall use standard icon according to IEC 60417-5009
-
The force required to activate the ON/OFF button shall be between 3.1 – 22.2 N (0.7 – 5 pounds)
Want to know more?
Insights you might also be interested in:

Why you should apply virtual DoE to the design process
Early product insights and fewer physical tests are some of the benefits of using Design of Experiments in medical device development.