Implementing an effective Risk Management process

Make the Risk Management process work for you with a few tips for smarter documents and procedures.
Philips Healthcare IntelliSave AX700

A full Risk Management process was developed for a new anesthesia workstation. We facilitated the development of documents and procedures to speed up the CE marking for the current device as well as future development projects.
Capillary Tube

We assisted with planning and documenting the usability engineering process according to IEC 62366 while at the same time laying out procedures and documentation for future products approvals.
ScanClime Humidity Control

A complex product redesign for efficient assembly and customization, which reduced production times by more than 25%.
Radiometer Medical ABL90
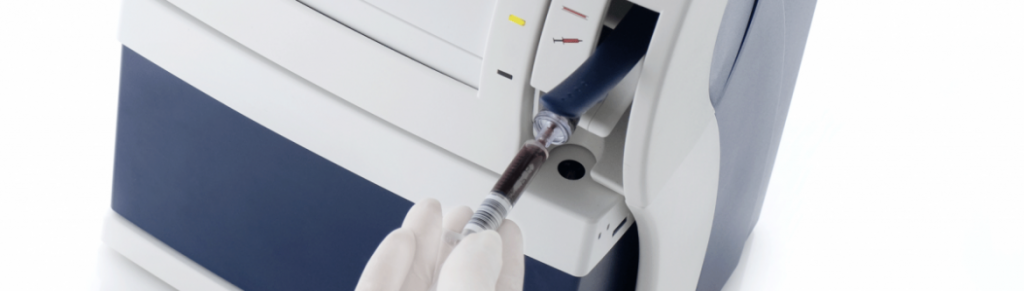
New product platform, new sensor technology, low cost production and high reliability – our contribution to concept development and product architecture helped to reduce response time and sample size by 50%.
Jabra Sport Pulse Wireless

In only three weeks we created a 3D master model of an in-ear heart rate monitor with automatic adjustment of subcomponents and near-scientific precision in measurements.
Axlab BiopSafe

An idea for a novel device was taken through the full product development process from idea to market launch, including setting up production, post-market surveillance, and a quality management system.
Ambu UltraSeal

We took on project lead in the development of a family of disposable face masks for anesthetic departments. The revolutionary design ensures easy use as well as individual fit and pressure setting.
SpeediCath Compact Set®

Driven by user needs for discreet appearance and easy portability, this award-winning catheter design is the result of systematic usability and mechanical engineering processes.
Good Relationships

We also praise our clients for the good things they do